Introduction
With the advent of modern multiple myeloma (MM) therapies, a significant proportion of patients can achieve durable clinical remission. Minimal residual disease (MRD) reflects depth of response and is a powerful predictor of progression-free survival (PFS) (Munshi et al, JAMA Oncology 2017). Although sustained MRD negativity (defined by IMWG as MRD-negative on two occasions, minimum one-year apart) is associated with improved PFS, there is continued long-term relapse in these patients. A recent study reported a 10-year PFS of 88% in MM patients who are MRD-negative > 3 years after total therapy (Mohan et al, Blood Advances 2022). In the current era of regular MRD testing in MM care, we aimed to estimate the 10-year PFS of unselected MM patients who are MRD and imaging-negative ≥ 5 years after latest therapy, using our single-center data.
Methods
MRD testing was introduced for MM patients in remission at the Icahn School of Medicine at Mount Sinai (ISMMS) in 2017. Therefore, we systematically reviewed all patients with MRD results in the ISSMS MM registry between July 2017 and December 2022. Next-generation multi-parametric flow cytometry (NGF) was used to detect MRD from 2017 to 2021, with the addition of next-generation sequencing (NGS) from 2021 onwards. MRD-negativity was defined as <1 in 10 5 by NGF or <1 in 10 6 by NGS, and one representative MRD result per year was taken for analysis. Whole-body positron emission tomography (PETCT) and MRI were used to evaluate bone or extra-medullary (EM) disease. Detailed retrospective chart review of patients with sequential MRD-negative results was conducted to identify those with durable clinical, MRD, and imaging negative remission. PFS was evaluated using Kaplan-Meier curves and survival estimates.
Results
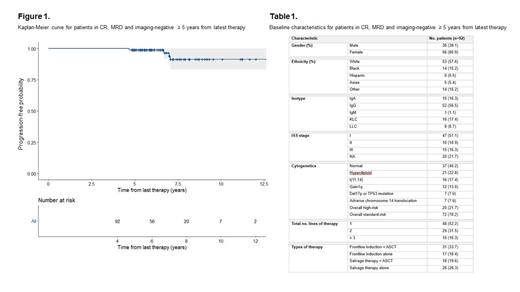
Between 2017 and 2022, 1703 MM patients underwent MRD testing at our center. Of these patients, we have so far identified 92 patients who were in CR, MRD and imaging-negative ≥ 5 years after the start of latest therapy.
The estimated 10-year PFS in our cohort was 91.4% (95% CI [0.833 ,1]; Figure 1). With a median follow-up time of 5.9 years (4.6 to 12.6 years), the majority of our cohort (75%) were off-therapy at last follow-up with a median time-off-therapy of 2.5 years. In terms of baseline patient and disease characteristics (Table 1), 57.6% patients were white and 60.9% were male. Most patients were ISS stage I (51.1%) with standard-risk cytogenetics (78.2%) at diagnosis. A much smaller proportion of our cohort had any adverse cytogenetics, including gain1q (13%) and del17p or TP53 mutation (7.6%). The majority of patients were in their first remission after frontline therapy (52.2%), with 33.7% having received autologous stem cell transplant (ASCT) first-line. A smaller proportion of the cohort had received salvage treatment; 31.5% had received two lines, 16.3% had received ≥ 3 lines.
Four patients relapsed in this cohort (4.3%) and one patient died of a non-MM related cause. Of the patients who progressed, 2 relapsed with isolated biochemical disease and 2 with predominantly EM disease. Both biochemical relapses were preceded by conversion to MRD positivity by NGF 6 months to 1 year prior, whereas those relapsing with EM disease remained MRD negative. Three patients had been off-treatment between 1 to 4 years prior to clinical relapse and the fourth patient progressed whilst on maintenance treatment.
Conclusion
We report single-institution MRD data on unselected MM patients who have durable MRD and imaging-negative remission for at least 5 years from their latest line of therapy. The estimated 10-year risk of disease-progression in our cohort is around 8.6%, with the majority of patients being off-therapy. The low rate of disease-progression at 10 years demonstrates that a proportion of MM patients do not suffer multiple relapses and may therefore be potential candidates for discontinuation of maintenance. From our data, these patients are more often ISS stage I, have standard risk cytogenetics and achieve deep responses to frontline therapy. A smaller proportion of patients are able to achieve this landmark even after salvage therapies. This may be increasingly possible in the era of monoclonal antibodies and T cell redirection therapies. Analysis of larger real-world MM cohorts is needed to elucidate whether sustained MRD-negative remission at 5 years can truly predict long-term disease-free survival and possible cure.
Disclosures
Richter:Adaptive Biotechnologies: Membership on an entity's Board of Directors or advisory committees; Astra Zeneca: Membership on an entity's Board of Directors or advisory committees; Abbvie: Consultancy; Janssen: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Genentech: Consultancy; Pfizer: Consultancy; Karyopharm: Membership on an entity's Board of Directors or advisory committees; Bristol-Meyers-Squibb: Membership on an entity's Board of Directors or advisory committees; Sanofi: Membership on an entity's Board of Directors or advisory committees; Takeda: Consultancy, Membership on an entity's Board of Directors or advisory committees. Cho:Takeda, Inc.: Research Funding; Bristol Myers-Squibb: Research Funding. Sanchez:Janssen Pharmaceuticals: Consultancy, Honoraria. Rodriguez:Janssen, Takeda, Bristol Myers Squibb, Amgen, Karyopharm Therapeutics: Membership on an entity's Board of Directors or advisory committees. Rossi:JNJ: Membership on an entity's Board of Directors or advisory committees; BMS: Membership on an entity's Board of Directors or advisory committees; Sanofi: Membership on an entity's Board of Directors or advisory committees; Adaptive: Membership on an entity's Board of Directors or advisory committees. Richard:Bristol Myers Squibb: Honoraria; C4 Therapeutics: Research Funding; Heidelberg Pharma: Research Funding; Janssen: Honoraria. Parekh:Amgen: Research Funding; Celgene/BMS Corporation: Research Funding; Karyopharm Therapeutics: Research Funding; Caribou Biosciences: Research Funding; Grail, LLC: Membership on an entity's Board of Directors or advisory committees. Jagannath:Legend Biotech: Consultancy; Karyopharm: Consultancy; Sanofi: Consultancy, Membership on an entity's Board of Directors or advisory committees; Regeneron: Consultancy; Bristol Myers Squibb: Consultancy; Caribou: Consultancy; Janssen: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal